Heart Failure
Heart failure is often driven by dysfunction of the heart muscle cell, or cardiomyocyte. These cardiomyocytes undergo large deformations at high speeds for ~2 billion contractile cycles over the lifetime of an organism, experiencing a swirling mix of stress, strain, torsion and shear. Their adaptation to this mechanical stress (or lack thereof) can determine disease progression, making them a fascinating, yet evasive subject for mechanobiological study.
Our research program probes looming questions on the forces of the heart. 1) How does subcellular architecture shape the mechanics of the heartbeat? 2) How does a heart cell sense and respond to changing external forces? 3) And critically, how does this all go wrong in heart failure?
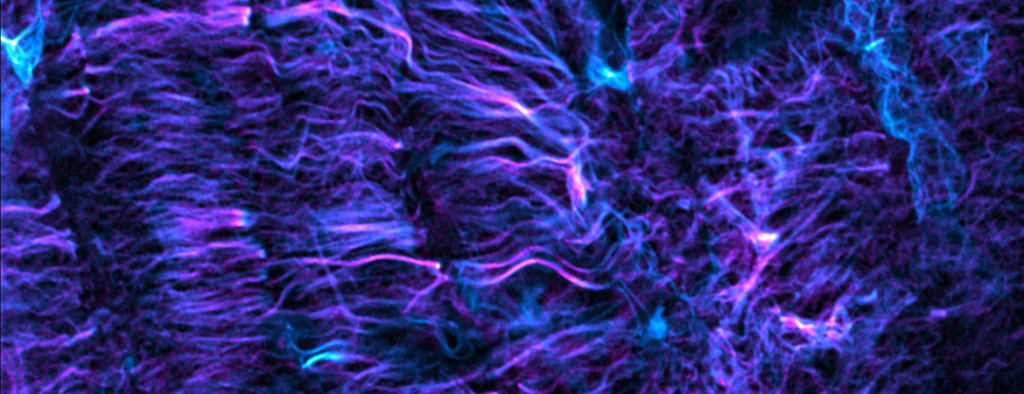
Several efforts stem from our unexpected discovery that microtubules act as load-bearing elements to regulate the mechanics of the heartbeat (video). This behavior is tunable, altered in human heart disease, and appears targetable for the treatment of heart failure. With vital basic and translational implications, we aim to precisely define this cytoskeletal contribution to cardiac mechanics, and to determine whether targeting microtubules holds therapeutic potential for currently untreatable forms of heart failure.
Yet to effectively target these cytoskeletal elements in heart failure, we must also better understand their basal functionality. Microtubules regulate essential homeostatic processes – how these are compromised in heart failure is unclear and requires a deeper understanding of their normal roles. We are pursuing the causes, consequences, and therapeutic targeting of this cytoskeletal remodeling as part of the Leducq Cytoskeletal Network (see photo) – a Trans-Atlantic Network of 7 expert, collaborating labs from the U.S., Germany, France, the Netherlands, and Israel, and with whom we are incredibly fortunate to work.
We are also advancing our translational program through multiple industry partnerships focused on developing novel small molecules and gene therapeutic approaches to correct aberrant microtubule remodeling in human heart failure.
Microtubule “buckling” in the cardiomyocyte